Dosage and Administration :
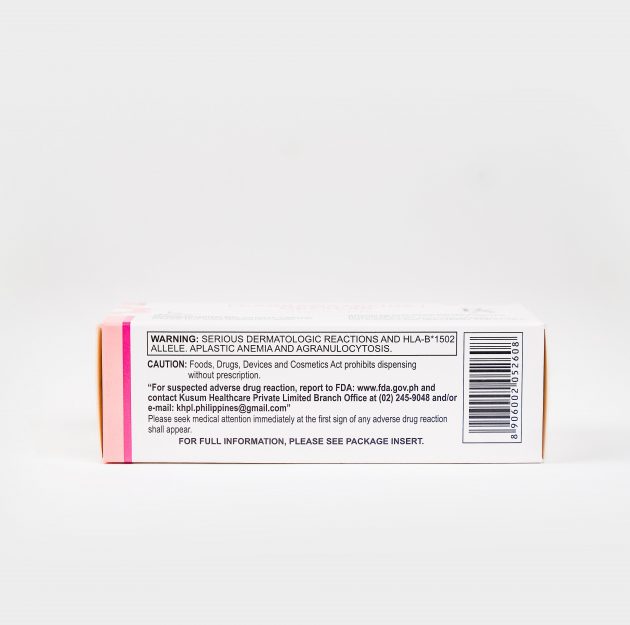
Before deciding to initiate treatment, patients of Han Chinese and Thai origin should whenever possible be screened for HLA-B*1502 as this allele strongly predicts the risk of severe carbamazepine associated Stevens-Johnson syndrome.
Epilepsy: The dose of carbamazepine should be adjusted to the needs of the individual patient to achieve adequate control of seizures. Determination of plasma levels may help in establishing the optimum dosage. In the treatment of epilepsy, the dose of carbamazepine usually requires total plasma-carbamazepine
concentrations of about 4 to 12 micrograms/mL (17 to 50 micromoles/litre) (see special warnings and precautions for use).
Adults: It is advised that with all formulations of Mezacar, a gradually increasing dosage scheme is used and this should be adjusted to suit the needs of the individual patient.
Mezacar should be taken in a number of divided doses although initially 100-200 mg once or twice daily is recommended. This may be followed by a slow increase until the best response is obtained, often 800-1200mg daily. In some instances, 1600 mg
or even 2000 mg daily may be necessary.
Elderly: Due to the potential for drug interactions, the dosage of Mezacar should be selected with caution in elderly patients.
Children and adolescents: It is advised that with all formulations of Mezacar, a gradually increasing dosage scheme is used and this should be adjusted to suit the
needs of the individual patient.
Usual dosage 10-20 mg/kg bodyweight daily taken in several divided doses. Mezacar tablets are not recommended for very young children. 5-10 years: 400 to 600 mg daily (2-3 x 200 mg tablets per day, to be taken in divided doses).
10-15 years: 600 to 1000 mg daily (3-5 x 200 mg tablets per day, to be taken in several divided doses). >15 years of age: 800 to 1200 mg daily (same as adult dose).
Maximum recommended dose
Up to 6 years of age: 35 mg/kg/day
6-15 years of age: 1000 mg/day
>15 years of age: 1200 mg/day.
Wherever possible, anti-epileptic agents should be prescribed as the sole antiepileptic agent but if used in polytherapy the same incremental dosage pattern is advised.
When Mezacar is added to existing antiepileptic therapy, this should be done gradually while maintaining or, if necessary, adapting the dosage of the other antiepileptic(s) (see interactions with other medicinal products).
Trigeminal neuralgia: Slowly raise the initial dosage of 200-400 mg daily until freedom from pain is achieved (normally at 200 mg 3-4 times daily). In the majority of patients a dosage of 200 mg 3 or 4 times a day is sufficient to maintain a pain free state. In some instances, doses of 1600 mg Mezacar daily may be needed.
However, once the pain is in remission, the dosage should be gradually reduced to the lowest possible maintenance level. Maximum recommended dose is 1200 mg/day. When pain relief has been obtained, attempts should be made to gradually
discontinue therapy, until another attack occurs.
Elderly
Dosage in Trigeminal neuralgia: Due to drug interactions and different antiepileptic drug pharmacokinetics, the dosage of Mezacar should be selected with caution in elderly patients.
In elderly patients, an initial dose of 100 mg twice daily is recommended. The initial
dosage of 100 mg twice daily should be slowly raised daily until freedom from pain is achieved (normally at 200 mg 3 to 4 times daily). The dosage should then be gradually reduced to the lowest possible maintenance level. Maximum recommended dose is 1200 mg/day. When pain relief has been obtained, attempts
should be made to gradually discontinue therapy, until another attack occurs.
For the prophylaxis of manic depressive psychosis in patients unresponsive to lithium therapy